 |
| New regulations outline the path towards more brand-name drugs being available across the country. Photo: Le Toan |
According to a VIR source, a new draft circular on drug tendering is on the table of leaders at the Ministry of Health for review before being issued in the next few weeks, meaning that multinational corporations (MNCs) like AstraZeneca Vietnam, Pfizer, GSK, and Novartis will experience the new change.
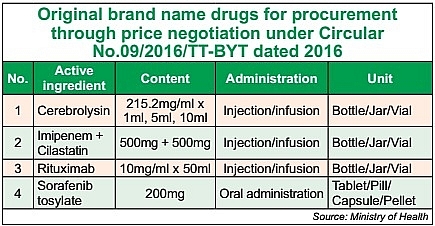
To replace 2016’s Circular No.09/2016/TT-BYT governing the list of drugs for procurement through bidding, for concentrated procurement, and for procurement through price negotiation, the new circular will expand the third list to drop the price of brand-name drugs.
Nitin Kapoor, chairman cum general director of AstraZeneca Vietnam, told VIR, “In light of these new rules, we continue to advocate for reasonable price negotiation as a fair mechanism to ensure patient access to innovative medicines and treatments on the market, as well as for an open business environment and policy predictability to make our investments and talent development programmes sustainable.”
Like AstraZeneca, Pfizer, GSK, and other members of the European Chamber of Commerce in Vietnam’s (EuroCham) Pharma Group – which represents the voices of 22 MNCs – support price negotiation, saying that public hospital drug tenders currently contribute more than two-thirds of the total prescription drug market for treatments, an exceptionally large number compared to other countries.
 |
“For this reason, in Vietnam, abrupt changes to tender policies will cause extensive impact to all actors in the market, and create a serious risk that patients and doctors will no longer be able to access innovative medicines, whether under reimbursement or self-pay,” said a Pharma Group representative.
“Implementation of price negotiation as based on existing regulations will not only ensure patient access and treatment optionality for doctors, but will also provide and maintain a predictable investment environment,” the representative added. “Reversing the course on price negotiation policy will jeopardise past, ongoing, and future investments of foreign and domestic companies.”
Aiming to increase local access to quality medicines and to reduce prices, focusing on brand-name drugs in line with the government’s direction, the new circular will add more branded products to the third list for procurement through price negotiation.
Risks come for MNCs as under the new circular there are two schemes for procurement of brand names: price negotiation, and open bidding in Group 1 under Circular No.15/2019/TT-BYT dated last July regulating drug tenders at public hospitals. Specifically, brand-name pharmaceuticals with many generic drugs in replacement in Group 1 will be included in open bidding for Group 1 drugs, while brand-name drugs failing in procurement via price negotiation will be also included in tender of Group 1.
The new rules should take effect in September, hoping to increase local access to quality and affordable prices of brand-name drugs. According to statistics from the Drug Administration of Vietnam, such products make up an average 26 per cent of total health insurance spending. The rate is 47 per cent at central hospitals, and 26 per cent at provincial ones.
Vietnam’s pharmaceutical industry, which in terms of market size is still at the emerging stage with an estimated total value of $7 billion in 2019, is growing at a robust pace of 8 per cent from 2019 to 2024 and is attractive to MNCs. Witnessing the growth potential, the groups are continuing to expand in the Southeast Asian country.
Building on the investment announcement of VND5 trillion ($220 million) last year during Prime Minister Nguyen Xuan Phuc’s visit to the global manufacturing site, AstraZeneca Vietnam recently signed a pharmaceutical distribution partnership with National Phytopharma JSC, making it one of the first of this model between a pharmaceutical MNC and a local distribution partner.
“It is also our next step to expedite our innovative solutions to address local healthcare needs with high quality, affordable medicines and contributing to developing the medical care and treatment for patients and communities,” noted Kapoor of AstraZeneca.
Similarly, Novartis Vietnam Ltd. on July 21 officially kicked off a public-private partnership project on strengthening primary healthcare project in the central province of Ha Tinh. This marked a new milestone on par with the MoU signed between the MoH and Novartis in 2019 to strengthen primary healthcare in Vietnam, towards universal health coverage and the sustainable development goals of the United Nations. VIR
Bich Thuy

VN pharmaceutical market: stiff competition fosters M&A wave
Vietnam ranks 17th among 'pharmerging' markets, or markets expected to become a ‘pillar’ for the world’s pharmaceutical industry, according to IMS Health.

Medical equipment, pharmaceutical companies see bright future after Covid-19
Most medical equipment and pharmaceutical companies reported high revenue and profit growth rates in Q1 in comparison with the same period last year.