 |
|
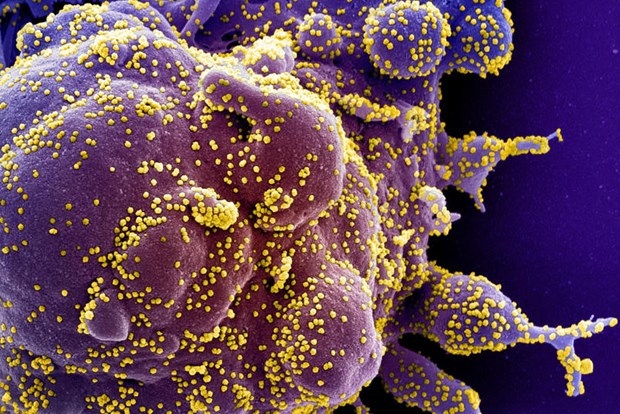
Colorized scanning electron micrograph of a dying cell (purple) infected with SARS-COV-2 virus particles (yellow), isolated from a patient sample. Image captured at the NIAID Integrated Research Facility in Fort Detrick, Maryland. (Photo: NIH via Reuters)
|
The announcement follows an initial cooperation agreement between the Thai Ministry of Public Health and Chinese pharmaceutical companies in developing a vaccine against the SARS-CoV-2.
Speaking at a meeting of the National Vaccine Committee, DDC chief Suwannachai Wattanayingcharoenchai said the National Vaccine Institute is preparing to sign a Memorandum of Understanding (MoU) with Chinese partners to test a COVID-19 vaccine in Thailand.
The trial is expected to involve more than 10,000 Thai citizens, he said, adding that DDC will consider possible candidates to work with.
According to Suwannachai, many laboratories in the country are ready to develop a vaccine if their Chinese counterparts provide them with the technology.
Public Health Minister Anutin Charnvrirakul said he has ordered the National Vaccine Institute to complete its vaccine development blueprint within three months so the country can go ahead with vaccine development projects.
The government has approved 45 billion THB for the fight against COVID-19, which included vaccine development, he noted.
Thailand on April 22 recorded 15 new cases of COVID-19, raising the total to 2,826, including 49 deaths.
The number of infections reported by day in Thailand is on the decline, opening up the possibility that the government may ease restrictions across the country in the coming time./.VNA
 Human trials of a COVID-19 vaccine in Thailand are expected to begin within the next four months, according to the Department of Disease Control (DDC) of the country.
Human trials of a COVID-19 vaccine in Thailand are expected to begin within the next four months, according to the Department of Disease Control (DDC) of the country.