- © Copyright of Vietnamnet Global.
- Tel: 024 3772 7988 Fax: (024) 37722734
- Email: evnn@vietnamnet.vn
nanogen
Update news nanogen
Deputy Minister of Health talks about fate of made-in-Vietnam Covid-19 vaccines
Deputy Minister of Health Do Xuan Tuyen said all three vaccines developed by Vietnam’s companies, including Nanocovax, Covivac and ARCT-154, are under clinical trials.
Health Ministry urges speeding up of locally made Covid-19 vaccines
After an emergency meeting, the National Ethics Council agreed to test the locally-made Covid-19 vaccine Nanocovax on 13,000 volunteers. This third phase trial will be completed in mid-August.
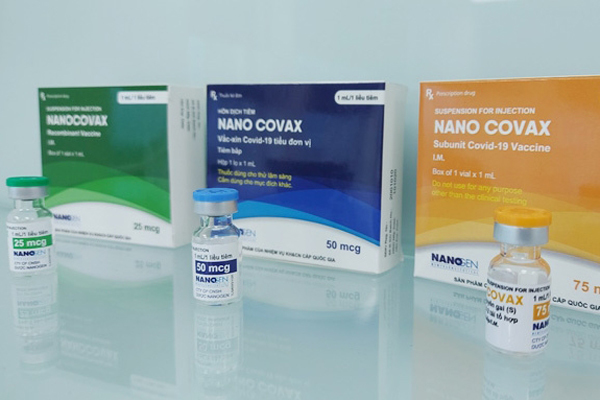
Made-in-Vietnam Nanocovax is expected to be as good as foreign vaccines
The test of locally-made Nanocovax vaccine has yielded positive signs. In case of emergency approval, Vietnam would be able to produce 100 million doses per year.
Vietnam may grant emergency license for locally-made Covid-19 vaccine
Soon after the phase 3 trial, Vietnam may consider emergency licensing for Nanocovax, a made-in-Vietnam Covid-19 vaccine.
Vietnam to have its first Covid-19 vaccine
Results of a clinical trial phase 2 of made-in-Vietnam Covid-19 vaccine Nanocovax showed that 100% of vaccinated people made antibodies to fight the virus. The vaccine was also effective on the British variant of the coronavirus.
Vietnam to produce Covid-19 vaccines by September
The clinical trial of the domestically-made Nanocovax vaccine is going smoothly, and by the end of September, Vietnam will have its first Covid-19 vaccine.
Vietnam's Covid-19 vaccine completes 50% of phase 2 trial
Vietnam has completed the first-dose vaccination of 560 volunteers who participated in the Phase 2 trial of Nanocovax's Covid-19 vaccine,
Volunteers get 2nd shot of 25mcg dose of Nanocovax
Three volunteers getting the first shot of the 25mcg dose of Nanocovax, the first Vietnamese COVID-19 vaccine to be tested on humans, last December received the second on January 14.
Human trails of 2nd local COVID-19 vaccine expected in January
The Institute of Vaccines and Medical Biologicals (IVAC), based in central Khanh Hoa province, has proposed the Ministry of Health allow human trials of its COVID-19 vaccine to begin in January, two months ahead of schedule.
Additional 17 volunteers injected with Vietnamese COVID-19 vaccine
A further 17 people out of the 60 volunteers participating in the first phase of human trials to develop Nanocovax, a locally-produced novel coronavirus (COVID-19) vaccine, received their vaccine shots on December 22 at the Military Medical Academy.
Made-in-Vietnam biopharmaceuticals produce stir on world market
VietNamNet Bridge - The heads of the Ministry of Health had doubts that the producer Nanogen had made a special drug -- until they saw how it was made.